Not all batteries are made equal. Also well known that alkaline batteries are beating traditional zinc-carbon batteries in terms of output power and capacity. But how are really good these batteries? And are alkaline batteries better than their rechargeable counterparts?
Let's test some set of AA type alkaline batteries side by side with Ni-MH rechargeable batteries of same type (Sanyo Eneloop 1800mAh and Varta Ready2Use 2100 mAh). To measure discharge characteristics we will use well known IMAX B6 intelligent charger connected to Raspberry Pi single board computer.

All batteries are discharged by constant current (200mA) down to 0.8V. And below are discharge curves:
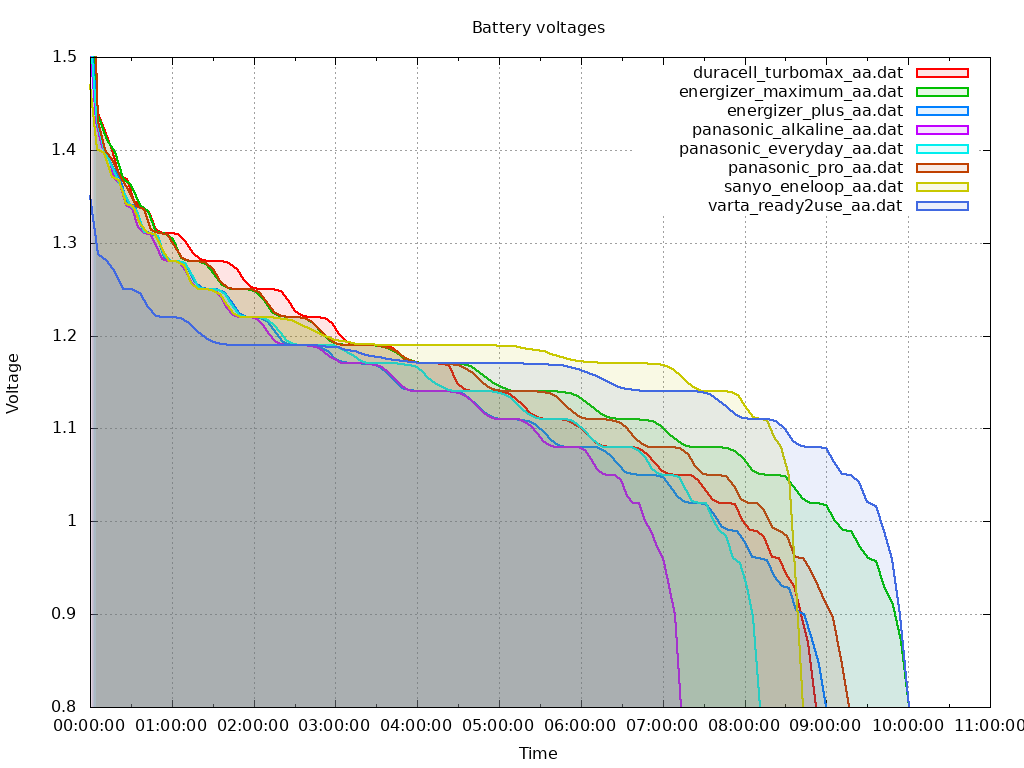
So alkaline batteries results are not so good as they are promoted by vendors! Despite to higher voltage in beginning (1.6...1.75V vs 1.3...1.45V), under load some time later alkaline battery voltage is falling below voltage of rechargeable battery.
Battery capacitance could be calculated from area under the discharge curve by integration, and multiplied by discharge current will result in total outputted energy (in joules):
| Battery | Energy, J | Capacity, mAh |
|---|---|---|
| Panasonic Alkaline | 6046.96 | 1442 |
| Panasonic Everyday | 6830.7 | 1637 |
| Energizer Plus | 7328.21 | 1798 |
| Duracell Turbomax | 7395.7 | 1773 |
| Sanyo Eneloop | 7513.83 | 1743 |
| Panasonic Pro | 7703.3 | 1853 |
| Energizer Maximum | 8310.02 | 2001 |
| Varta Ready2Use | 8310.19 | 2000 |
Last column is capacity of battery measured by charger (in mAh).
So, we can make some conclusions:
- Alkaline batteries are not so great. Only expensive ones have capacitance comparable to their rechargeable counterparts.
- More mAh isn't more capacity in terms of total energy (compare Duracell Turbomax and Sanyo Eneloop results).
- Alkaline batteries are well suited for low consumption devices (like remote controls, clocks, etc) and for extended temperature range devices (like outdoor sensors). If your device is high current and is not subject of low temperatures - rechargeable Ni-MH battery is your choice.
Some words about methodics
Charger data was logged by modified Niobos perl script running under Raspbian Linux. USB port was switched to 9600 8N1 raw mode using stty command:
$ stty -F /dev/ttyUSB0 cs8 -cstopb -ixon raw speed 9600
Text log file was created while battery discharging by command
$ b6_decode.pl < /dev/ttyUSB0 | tee out.txt
Resulting .txt files were transformed to .dat files with layout suitable to drawing graphs using gnuplot utility. Outputted energy was calculated by estimate_energy.sh script.
If you have IMAX B6 charger connected to Linux box, you could make your own experiments with batteries using mentioned scripts downloaded here.

Comments
I wonder how did you reach the conclusion about alkaline batteries as being better suited for low consumption devices. Granted, this is quite true, however this fact can certainly not be based upon the experiment described in your article. Have you examined the capacities of various types of batteries under various types of load? Obviously not. Second,the calculation of the integral of the voltage discharge curve will not give you the charge capacity. Why would it? Total nonsense. I suggest to You a more careful approach in publishing experiments and the conclusions that may follow. Programming and hardware inter-phasing is a very excellent skill to have,but basic theory on a scientific level is equally important to master. Or at least to manage.
Emery Polan thank you for your comment, these moments definitely need a better explanation. Alcaline batteries are more suitable for low consumption devices because of high self-discharge rate of most Ni-MH batteries (low self-discharge Ni-MH batteries are out of scope of this article). And you're right, the calculation of the integral of the voltage discharge curve by itself doesn't make any sense, but the integral of the voltage multiplied by discharge current does. Since the discharge current is constant, it can be separated from the integrand and instead multiplied by the integral of the voltage discharge curve. You could check out full set of sсripts used for capacity calculations for additional information.
-Yes, I saw that the second part of that sentence relating to the total energy is correct, and I also immediately saw the reason for that as being equal to the power curve time integral when the constant current is brought back as a multiplier within the integrand. I commented on the total charge formula being correct as well, but under an "anonymous" entry. But all this is very basic, really. -There is a little more to why alkaline batteries perform better with low consumption, and it is not the low self discharge rate. These batteries simply have a highly variable capacity under different loads. Most AA alkalines will easily surpass the 3000 mah mark in low consumption applications, while can only muster 1/3 of that at the 1 or 2 ampere drain rate. If even that. So, even super low self discharge Nimh batteries will perform poorly when compared to alkalines in low drain applications. I think that in an article such as this one has to be a little more careful with the clarity of conclusions reached and the logic of making inferences from the experiment alone. Otherwise, what is the point of going into the detail of the entire setup and obtaining the data. Not to be critical, really just want to be constructive...